Intraoperative Radiation Therapy for Breast Cancer
 Tri-City Medical Center is one of the first hospitals in San Diego County, and the first in North San Diego County, to offer intraoperative radiation therapy (IORT). Combining surgery and radiation, this form of treatment is for treating certain types of breast cancer – it works by introducing a single dose of radiation therapy immediately after a lumpectomy. The goal of this revolutionary procedure is to avoid unnecessarily exposing healthy breast tissue to radiation for extended periods of time, thereby reducing side effects and the length of treatment.
Tri-City Medical Center is one of the first hospitals in San Diego County, and the first in North San Diego County, to offer intraoperative radiation therapy (IORT). Combining surgery and radiation, this form of treatment is for treating certain types of breast cancer – it works by introducing a single dose of radiation therapy immediately after a lumpectomy. The goal of this revolutionary procedure is to avoid unnecessarily exposing healthy breast tissue to radiation for extended periods of time, thereby reducing side effects and the length of treatment.
For most women diagnosed with early stage (Stage I) breast cancer, IORT may be the best treatment option to treat patients while retaining as much healthy breast tissue as possible.
Learn More By Calling
855.222.8262 Today!
Tri-City Medical Center
4002 Vista Way
Oceanside, CA 92056
Benefits of IORT
Compared to traditional radiation therapy or a mastectomy – the removal of the entire breast tissue – IORT employs combines radiation therapy and a lumpectomy – the removal of a portion of the affected breast tissue – which offers a variety of important benefits for women diagnosed with early-stage breast cancer.
Traditional radiation treatment is administered to the entire breast and flank of the body, and patients are required to visit the hospital five days per week for about six weeks. This is both a time burden and a health issue – a patient must put their lives on hold while undergoing treatment as well as experience the negative effects of radiation exposure.
In contrast, IORT delivers a single, highly-localized dose of radiation directly in the void removed during the lumpectomy. This offers a pinpoint treatment option for patients in a fraction of the time leading to quicker recovery as well as less of a health risk.
IORT Procedure

- Step 1 – The surgeon performs a lumpectomy, removing the cancerous tissue from the breast, creating a small cavity in the breast tissue.
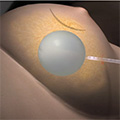
- Step 2 – The radiation oncologist inserts the balloon applicator into the cavity of the breast where the tumor was located.
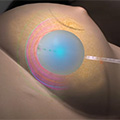
- Step 3 – Radiation is administered to the breast tissue through the balloon applicator; treatment is conducted in as little as eight minutes.
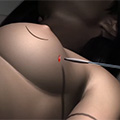
- Step 4 – The balloon applicator is removed and the surgeon sutures the cavity shut.
FAQs
What are the benefits?
Studies have shown IORT is as effective as traditional radiation treatments in treating early stage (Stage I) breast cancer. IORT also eliminates the need for multiple radiation treatments, reducing the negative health side effects of radiation treatment.
Does the treatment hurt?
Patients undergoing IORT will likely not experience side effects any differently than undergoing lumpectomy. Patients are put under anesthesia while the surgeon works to remove the tumor and surrounding breast tissue. After the surgeon completes the lumpectomy, the surgeon and radiation oncologist will administer the radiation treatment.
How long does the treatment last?
An IORT treatment will last anywhere between 30 and 60 minutes depending on the severity of the diagnosis.
Who is eligible for IORT?
IORT is available to patients diagnosed with early stage (Stage I) breast cancer and undergoing a breast lumpectomy.
Will I still require radiation treatments after undergoing IORT?
Some patients may be required to undergo traditional radiation therapy as a follow-up to ensure the treatment is effective. In this case, IORT is considered a “boost” to the subsequent treatments which can help reduce follow-up treatment times and eliminate the possibility of post-surgery complications.
Currently, IORT is available to breast cancer patients who undergo lumpectomy to remove breast tumors less than two centimeters in size.
References
Dickler, et al. “A Dosimetric comparison of MammoSite high dose rate brachytherapy and Xoft Axxent electronic brachytherapy,” Brachytherapy (6) 2007, 164–168.
Risk-adapted targeted intraoperative radiotherapy versus whole-breast radiotherapy for breast cancer: 5-year results for local control and overall survival from the TARGIT-A randomised trial. Lancet Volume 383, No. 9917, p603–613, 15 February 2014.
