THE MOST UP TO DATE TRI-CITY NEWS & HEALTH NEWS
Tri-City News
Tri-City Medical Center Community Update
Dear Community, On Thursday, November 9, 2023, Tri-City Medical Center became aware of unauthorized activity on our computer network. We immediately took our systems offline to halt the activity and engaged [...]
Exciting News: A Transformative Step Forward
Letter from the CEO: Tri-City Medical Center’s President and CEO: Dr. Gene Ma Dear Tri-City Community, I am thrilled to share with you some momentous news that we believe will mark [...]
DANDY LEE, M.D. – BELOVED PHYSICIAN, COLLEAGUE, MENTOR AND FRIEND
DANDY LEE, M.D.NOVEMBER 12, 1963 - MAY 24, 2023BELOVED PHYSICIAN, COLLEAGUE, MENTOR AND FRIEND Dr. Dandy Lee served at Tri-City Medical Center (TCMC) as an anesthesiologist for more than [...]
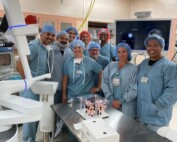
Ion Robotic-Assisted Bronchoscopy: New Technology Helps Physicians at Tri-City Medical Diagnose Lung Cancer Earlier
Ion Robotic-Assisted Bronchoscopy: New Technology Helps Physicians at Tri-City Medical Diagnose Lung Cancer Earlier Tri-City Medical Center (TCMC) now offers robotic-assisted bronchoscopy, a minimally invasive procedure used to biopsy [...]
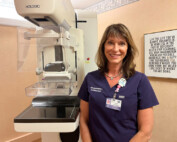
Tri-City Medical Center Offers Full Service of Imaging Tests for Early Detection of Breast Cancer
Jennifer Mayberry “Screening for breast cancer is one of the most important things a woman can do for her health,” said Jennifer Mayberry, MD, who specializes in in diagnostic [...]
Acute Rehabilitation at Tri-City Medical Center: Team Approach & Continuity of Care
Acute Rehabilitation at Tri-City Medical Center: Team Approach & Continuity of Care Rehabilitation services at Tri-City Medical Center include a 10-bed acute rehabilitation unit, inpatient services, and outpatient services [...]